Experiment Title: Single-Molecule Fluorescence Imaging Based on the AttosTek UVISI064BU High-Sensitivity Camera
Institution: Super-resolution Fluorescence Imaging Laboratory, National Center for Nanoscience and Technology
Experiment Date: November 2023
I. Application Background and Practical Significance
Single-molecule fluorescence imaging is a revolutionary technology in modern life sciences, nanotechnology, and materials science. By detecting fluorescence signals at the single-molecule level, it enables direct observation of biological molecule behavior, dynamic processes, and nanoscale structures. Super-resolution microscopy techniques based on single-molecule localization (such as PALM/STORM) break the diffraction limit of traditional optical microscopes, pushing spatial resolution to the nanometer scale.
Technical Challenge
Single-molecule fluorescence signals are extremely weak. Each molecule emits only a limited number of photons (typically a few hundred to a few thousand) per imaging cycle and is subject to continuous photophysical changes (e.g., blinking, photobleaching). Therefore, achieving high signal-to-noise ratio (SNR) and high temporal resolution in single-molecule detection imposes ultimate requirements on the core detector of the imaging system—the scientific camera: extremely high quantum efficiency, ultra-low readout noise, and fast, distortion-free electronic readout capability.
Significance of this Experiment:
The purpose of this experiment is to evaluate the practical performance of the AttosTek UVISI064BU high-sensitivity camera in the critical application scenario of single-molecule fluorescence imaging. By observing the single-molecule fluorescence patterns and dynamic blinking of the classic fluorescent probe Rhodamine 6G, we aim to directly verify the camera’s core capabilities in single-photon detection sensitivity, temporal resolution, and localization accuracy.
Promotion for Related Industries and Products:
- Biomedical Research and Precision Medicine:
Cell Biology and Neuroscience: High-sensitivity single-molecule imaging can resolve the nanoscale distribution, aggregation states, and dynamic interactions (e.g., receptor clustering, signal transduction) of proteins on cell membranes in real-time, providing dynamic molecular-level images for understanding processes like neuronal synapse formation, immune responses, and viral infection.
Pathological Diagnosis and Drug Development: It enables the observation of changes in the distribution of biomarkers at the nanoscale, offering new tools for studying protein aggregation abnormalities related to diseases such as early cancer diagnosis and Alzheimer’s disease. Simultaneously, it allows real-time tracking of drug molecule targeting, binding, and metabolism within cells, accelerating the development and screening of new targeted drugs.
- Materials Science and Nanotechnology:
- Nanomaterial Characterization: It can be used to study the luminescence uniformity, photostability, and surface modification effects of nanomaterials like quantum dots, upconversion nanoparticles, and fluorescent polymers, evaluating their performance at the single-particle level.
- Energy and Catalysis: By labeling active sites on catalyst surfaces, single-molecule events during catalytic reactions can be observed, revealing catalytic mechanisms.
II. Experimental Principle
This experiment is based on wide-field fluorescence microscopy combined with single-molecule detection and localization techniques.
- Single-Molecule Fluorescence Detection: The fluorescent probe (Rhodamine 6G) is diluted to an extremely low concentration (~1 nM) and immobilized on a substrate. This ensures that, on average, only one or fewer fluorescent molecules exist within the diffraction-limited area (approx. 250 nm diameter). Upon excitation by a specific wavelength laser (561 nm), a single molecule absorbs photons, transitions to an excited state, and subsequently emits fluorescence photons through radiative transitions.
- Point Spread Function (PSF) and Localization: Light emitted from a single point-like fluorescent source, after passing through the microscope objective, forms a two-dimensional Gaussian-like intensity distribution on the camera plane due to diffraction. This is known as the Point Spread Function (PSF). The camera’s task is to record this PSF with the highest possible signal-to-noise ratio.
- Fluorescence Blinking: Many fluorescent molecules (including Rhodamine 6G) can randomly enter a non-fluorescent dark state (e.g., triplet state) and later recover emission. This phenomenon is called “blinking”. Observing single-molecule blinking is crucial evidence for identifying individual molecules as opposed to clusters or impurities, and it is also a physical mechanism exploited by some super-resolution techniques.
- From Single Molecules to Super-Resolution: By continuously acquiring multiple image frames, recording the stochastic emission and localization processes of numerous single molecules, and then superimposing all these precisely localized positions (with accuracy far below the diffraction limit) onto a single reconstructed image, a structure of the sample with resolution far exceeding the diffraction limit can be obtained.
III. Task Description
Disperse and immobilize a low concentration of fluorescent probe molecules onto the surface of a coverslip. Under excitation light matching their absorption wavelength, use a highly sensitive camera to record the fluorescence patterns of individual fluorescent probes. Simultaneously, observe the temporal intensity fluctuations (blinking) of the fluorescence signal from single probe molecules.
IV. Test Equipment and Key Parameters
- Core Detector: AttosTek UVISI064BU High-Sensitivity Camera

Expected Key Performance: High quantum efficiency (especially in the visible range), low readout noise (< 1.5 e-), high full-frame readout speed.
- Imaging System: Inverted fluorescence microscope equipped with a high numerical aperture (NA) oil immersion objective.
- Excitation Source: 561 nm continuous-wave (CW) diode laser, used for exciting Rhodamine 6G.
- Fluorescent Probe: Rhodamine 6G, a common calibration standard for single-molecule fluorescence, with an excitation peak around 525 nm and emission peak around 550 nm.
- Sample: 1 nM Rhodamine 6G aqueous solution spin-coated onto a clean coverslip surface.
V. Experimental Procedure
- Sample Preparation: A drop of 1 nM Rhodamine 6G solution was placed onto a clean coverslip and spin-coated at an appropriate speed to create a sparse layer of surface-adsorbed single molecules. The sample was then air-dried.
- System Setup and Alignment: The sample was placed on the microscope stage. The optical path was equipped with the 561 nm laser excitation module, corresponding dichroic mirror, and emission filter. The light path was carefully aligned to ensure uniform illumination conjugated with the imaging path.
- Imaging and Data Acquisition:
- Appropriate exposure time (e.g., 10-50 ms), gain, and readout mode were set in the camera control software.
- A field of view with sparse, clearly visible fluorescent molecules was located.
- A continuous sequence of image frames was acquired to record the appearance, blinking, and photobleaching processes of single-molecule fluorescent spots.
VI. Experimental Results and Analysis
Experimental Results:
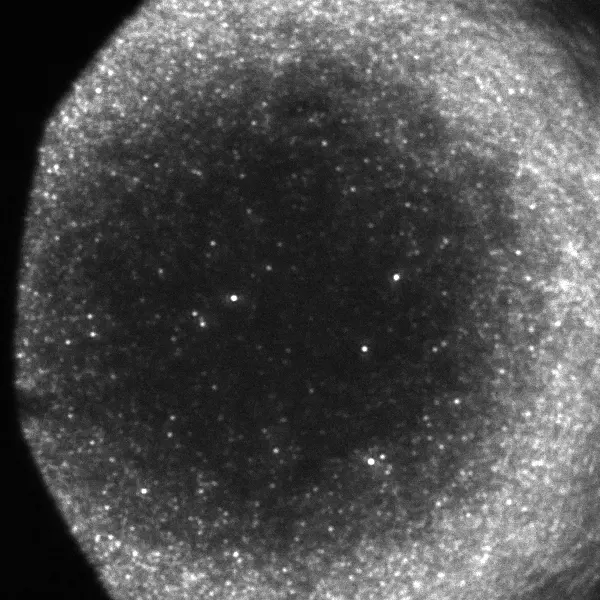
Result Analysis:
- Verification of Successful Single-Molecule Detection:
In a single image frame, multiple isolated, bright, approximately circular diffraction-limited spots can be observed. These spots are well-separated, consistent with the characteristics of a sparse single-molecule distribution. The signal-to-noise ratio (SNR) of the spots is high, with a uniformly clean background. This directly demonstrates the excellent sensitivity and ultra-low noise level of the UVISI064BU camera, confirming its ability to efficiently capture the limited number of photons emitted by a single fluorescent molecule within a short time.
- Observation and Analysis of Single-Molecule Characteristics:
“On-Off” Blinking Phenomenon: Temporal intensity trace analysis of individual fluorescent spots reveals characteristic random “on-off” fluctuations, i.e., blinking. This is a hallmark behavior of single molecules, distinguishing them from fluorescent clusters or autofluorescence. The camera’s fast frame rate ensures accurate capture of these rapid photophysical dynamics.
VII. Conclusion
This experiment successfully achieved clear imaging and dynamic observation of single Rhodamine 6G molecules using the AttosTek UVISI064BU high-sensitivity camera on a wide-field fluorescence microscope. The experiment not only observed isolated diffraction-limited spots characteristic of single molecules but also captured their hallmark fluorescence blinking.
Conclusions are as follows:
- Performance Validation: The AttosTek UVISI064BU camera’s performance in terms of single-photon detection sensitivity, temporal response, and low-noise control fully meets, and even exceeds, the stringent requirements of single-molecule fluorescence imaging and localization-based super-resolution microscopy techniques.
- Demonstration of Core Value: The high performance of this camera is the fundamental guarantee for successfully achieving single-molecule detection and holds promise for further enabling nanoscale super-resolution imaging. Its reliability as a core component has been validated.
